Diabetes device data sharing
There has been a significant investment in technology to improve the lives of people with diabetes. Devices that help people to monitor and manage glucose levels lead to improved quality of life and fewer diabetes emergencies.
However, many of the potential benefits of this technology remain untapped. Health professionals cannot easily or quickly access patient-generated data between or during clinic sessions, whether for direct clinical care or, as a digital safety net, to support patient-initiated follow-up and ensure those who need support are prioritised for review.
Currently, data is often shown to the healthcare professional on a smartphone by the person with diabetes or accessed through proprietary systems. However, if integrated with electronic health records, this data could enable healthcare professionals to more easily provide better personalised treatment.
There is a rapidly a growing number of devices and proprietary platforms but there are currently no internationally agreed interoperability standards to enable consistent data sharing.
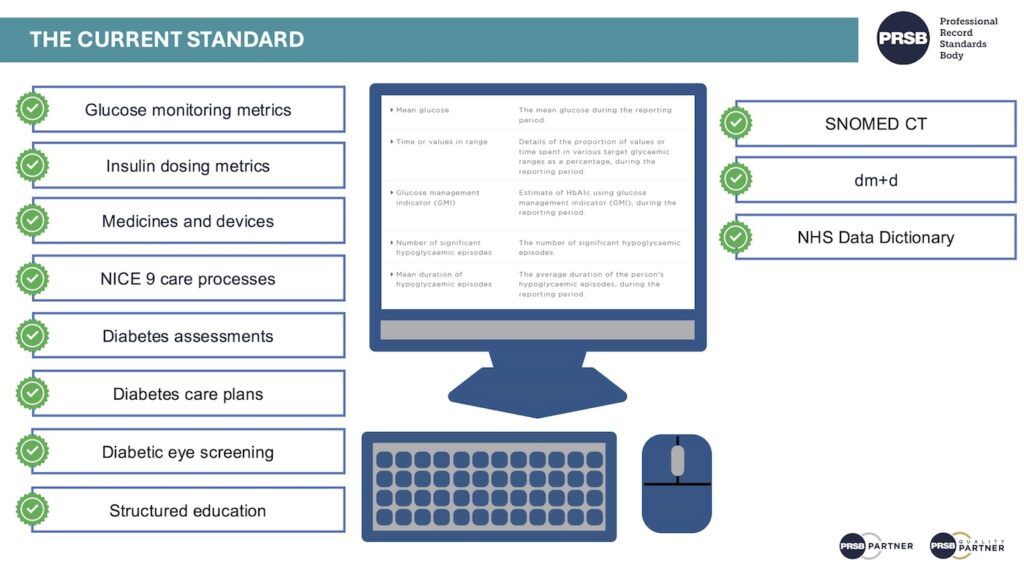
Background - The Diabetes Standard
Commissioned by NHS England and developed by PRSB with input from over 500 consultation participants, version 1.0 was published in 2023 with an Information Standards Notice (ISN) under section 250 of the Health and Social Care Act 2012.
The standard is endorsed by organisations and professional bodies including the Association of British Clinical Diabetologists, Diabetes UK, Royal Colleges of Emergency Medicine, General Practitioners, Nursing, Ophthalmologists, Psychiatrists, the Royal Pharmaceutical Society, the UK Kidney Association and the College of Podiatry.
The next release is planned for 2026 as part of this phase of work focused on patient-generated data from diabetes management devices.

Aims of next phase
This next phase of work will build on the existing record standard, enabling the standardised, safe, interoperable, and scalable sharing of patient-generated data from diabetes management devices across health and care settings in England.
The main focus will be to support clinicians to integrate patient generated data from diabetes management devices into routine care safely and efficiently. We will also explore how this data can be used to identify people with a higher clinical risk, so that clinical resources are directed to those who need support most, particularly where capacity is limited, and services can be redesigned around a more proactive, risk based approach.
Throughout this work, we will collaborate with healthcare professionals, the Association of British HealthTech Industries, industry partners, Diabetes UK, and international standards initiatives to develop solutions that support the sharing of patient-generated data. This data may be captured from Continuous Glucose Monitors (CGM), insulin pumps and connected insulin pens and Hybrid Closed Loop/ Fully Closed Loop systems (which are automated insulin delivery systems: including sensor glucose readings, insulin delivery events and information about the algorithm used for calculation).
Areas of focus
1. Information models that align with the latest international clinically-validated consensus guidance for insulin and glucose metrics (e.g. time in range)
2. Scalable architectural options to support implementation of diabetes device data sharing across the NHS.
3. HL7 FHIR resources to support the sharing of data from diabetes devices based on international standards.
4. Information Governance and medico-legal guidance to provide clarity on lawful bases under UK GDPR and the common law duty of confidentiality for processing device data for direct care and population health management. Guidance will address medico-legal concerns, including clinician liability where device data is accessible outside routine appointments, clarification of expectations regarding active monitoring versus availability of data and risk management approaches and patient communications.
5. Consensus agreement for the consistent display of data supporting clinicians in safely and effectively using device data.
6. Identification of pilot areas for testing and evaluating the outputs from this work.
Consultation
We will be seeking input as part of the consultation process and working with stakeholders to inform development of this next phase of the standard. Information and invitations to get involved will be shared as the work progresses.
Please follow our social media channels and visit this page for updates. If you are interested in contributing to this work, please complete the form below or contact us at info@theprsb.org.